Building E-E-A-T in Beauty & Personal Care Devices (2025): Certifications, Specs, and Compliance Pages That Win Trust
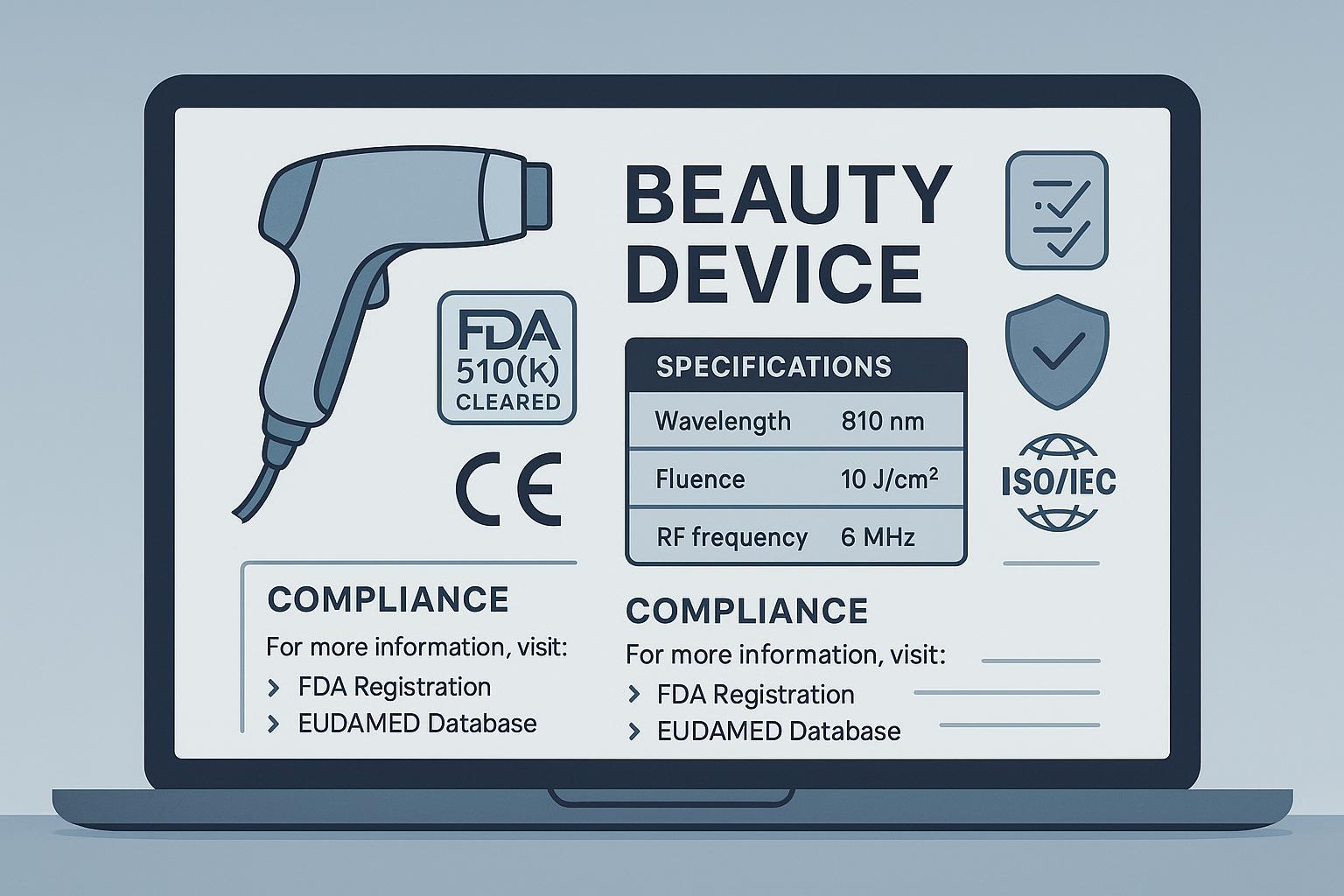
If you sell beauty or personal-care devices, you live in a Your Money or Your Life category. In 2025, trust isn’t just a brand value—it’s table stakes for search visibility and conversion. The fastest way I’ve seen device brands earn Experience–Expertise–Authoritativeness–Trust (E‑E‑A‑T) is by turning their certifications, technical specifications, and compliance documentation into clear, verifiable on-site proof. This guide distills what consistently works across US/EU/UK launches and audits, and how to ship it without derailing marketing momentum.
Key idea: don’t tell users and Google you’re trustworthy—show them with verifiable evidence, precise claims, and living compliance pages.
1) E‑E‑A‑T for device brands in 2025: what actually matters
- People-first quality: Google’s quality documentation continues to emphasize showing experience, expertise, and trustworthy sourcing on YMYL pages. If you make health-adjacent claims, expect higher scrutiny. See Google’s guidance in “Creating helpful, reliable, people‑first content” (Google Developers, updated 2024–2025) at Google helpful content guidance and the current Google spam policies.
- Proof beats prose: Device buyers look for safety, efficacy, and proper usage. Publish the exact specs, testing standards, and regulatory status, and link to primary registries where possible.
- Avoid claim creep: Cosmeceutical language that implies treatment or structure/function effects can push a product into drug or device territory in the US. When a device is involved, claims must align with its actual regulatory pathway and evidence.
Trade-off to manage: marketing flair vs. precision. If a slogan can be misread as a medical claim, it will be—by both consumers and regulators.
2) Certifications and regulatory status: what to say (and not say)
United States
- Use the right verbs:
- “FDA approved” is generally reserved for PMA Class III devices and drugs. Most consumer beauty devices, if regulated, are Class I/II and are not “approved.”
- “FDA cleared” applies to devices with successful 510(k) clearance. You can invite verification through the FDA 510(k) database (FDA, ongoing).
- “FDA registered” refers to establishment registration/device listing. It’s not an endorsement. You can point to the FDA Registration & Listing database (FDA, ongoing) with neutral language.
- Wellness-only devices: Some low‑risk products marketed with general wellness claims may fall under enforcement discretion; keep marketing aligned with that scope and maintain evidence consistent with wellness claims. Align your language with FDA’s published policies and avoid implying diagnosis/treatment.
- Cosmetics on the same site: The Modernization of Cosmetics Regulation Act (MoCRA) introduced facility registration, product listing, safety substantiation, serious adverse event reporting, and upcoming fragrance allergen disclosure. Keep your transparency page current using the FDA MoCRA hub (FDA, updated 2024–2025) for deadlines and guidance.
Practical placement:
- On the product page, include a “Regulatory Status” block with exact pathway language (e.g., “510(k)-cleared for [indication], K######”) and a short disclaimer (“FDA registration does not imply approval”). Offer “Verify clearance” linking to the FDA 510(k) database entry.
European Union (plus Great Britain)
- EU MDR applies to medical devices; certain aesthetic products with no medical purpose—like lasers/IPL—fall under Annex XVI and must meet Common Specifications. The European Commission’s Implementing Regulation (EU) 2022/2346 sets these requirements; see Implementing Regulation (EU) 2022/2346 Common Specifications (EUR‑Lex, 2022) alongside the consolidated EU MDR 2017/745 text (EUR‑Lex, 2023).
- CE marking is for devices/electrical products—not for cosmetic formulations. Keep cosmetics under Regulation (EC) No 1223/2009; notify via CPNP (not publicly visible) and state that notification is completed. You can reference the Commission’s CPNP information page (European Commission, ongoing) for context.
- UK (Great Britain): UKCA is replacing CE with transitional acceptance timelines. See MHRA’s current guidance in Regulating medical devices in the UK (MHRA, updated 2024–2025).
Practical placement:
- Add your Notified Body’s ID and scope, and link to its entry in the NANDO database (European Commission, ongoing). For Annex XVI devices, list CS‑driven warnings/training requirements in plain language on EU PDPs and in IFUs.
Microcopy that prevents trouble:
- “Our manufacturing facility is registered with FDA; registration does not imply product approval.”
- “CE marking applies to devices; cosmetic products do not carry CE marks.”
3) Technical specifications that build trust (and reduce returns)
Your PDP should read like a spec sheet a clinician would respect, and a consumer can parse in seconds. What I include per device type:
- LED/Photobiomodulation masks: Wavelength(s) in nm, irradiance (mW/cm²) at treatment distance, treatment time, total dose per session (J/cm²), photobiological safety reference such as “Evaluated to IEC 62471; see user precautions.” Where applicable, add medical electrical safety references such as “Evaluated to IEC 60601‑1 and IEC 60601‑1‑2.” You can cite standard overviews like IEC 62471/60601 previews (IEC Webstore, current preview pages) when explaining scope in your knowledge center.
- Laser/IPL: Wavelength band(s), pulse width and fluence range, spot size, skin type compatibility (Fitzpatrick types tested), safety references such as “Evaluated to IEC 60825‑1” for laser products; see IEC 60825‑1 overview (IEC Webstore). For home-use IPL, reference applicable household/device standards in your documentation.
- RF/Microcurrent/EMS: Frequency (kHz/MHz), power/current ranges (A or mA), duty cycle, electrode/contact materials, body areas approved, contraindications. For home-use devices, reference usability/safety collateral like “Home‑use considerations per IEC 60601‑1‑11”; see IEC 60601‑1‑11 overview (IEC Webstore).
- Biocompatibility: List all skin-contact materials and test approach (e.g., “Evaluated for skin contact per ISO 10993 series”). An accessible explanation from an accredited CB is helpful for users; see Intertek’s overview on biocompatibility testing to ISO 10993 (Intertek, ongoing).
- Electrical and EMC: Input voltage, power, battery specs, charging time, EMC classification, operating/storage environment.
Format tips that consistently perform:
- A scannable “Key Specs” card at the top; a downloadable PDF spec sheet and IFU below.
- Pictograms and symbol legend aligned to ISO 15223‑1; keep a simple legend page users can reference. Maintain version/date control in the footer of PDFs.
4) Compliance and transparency pages: a living document, not a brochure
High‑performing compliance pages are specific, verifiable, and updated at least annually. Structure them by market:
-
United States
- Device regulatory status: Class, pathway (510(k)/De Novo/PMA), intended use/indications, links to the FDA 510(k) database or De Novo/PMA databases (FDA, ongoing) as applicable.
- Establishment registration and device listing: link to the Registration & Listing database (FDA, ongoing) with neutral language.
- Adverse events/recalls monitoring statement with links to MAUDE (adverse events) and device recalls (FDA, ongoing).
- Cosmetics (if sold): MoCRA status—facility registration, product listing, safety substantiation approach, and allergen labeling plans; link your summary to the FDA MoCRA hub (FDA, 2024–2025 updates).
-
European Union
- Device classification, CE marking basis, Annex XVI applicability, and compliance against Common Specifications; reference Implementing Regulation (EU) 2022/2346 (EU, 2022).
- Notified Body details and a link to its NANDO profile (European Commission, ongoing).
- Consumer-facing explanations of warnings/training where required by CS.
-
Great Britain
- UKCA/CE acceptance status and labeling; cite MHRA device regulation guidance (MHRA, updated 2024–2025).
Sitewide trust utilities to include in your footer and Compliance hub:
- Warranty and returns; complaint-handling workflow and contact.
- Privacy and data protection; UGC moderation and review policy.
- “Report a product issue” form linking to your safety/compliance team.
Keep a “Last updated” timestamp and change log. It signals active stewardship to both users and auditors.
5) Claims, clinical evidence, and expert review: how to show your homework
- Substantiate proportionally: Health-related claims must be truthful, not misleading, and supported by competent and reliable scientific evidence. The FTC’s 2023 “Health Products Compliance Guidance” clarifies expectations for study quality, typical results, and visuals; see the FTC’s health claims guidance (FTC, 2023) and the updated Endorsement Guides (FTC, 2023) for testimonials/influencers.
- Publish a “Clinical Evidence & Studies” section on each PDP:
- Brief study summaries (design, population, endpoints, limitations).
- Links to peer‑reviewed publications where available.
- A plain‑English interpretation that avoids overgeneralizing.
- Expert review and authorship:
- Display reviewer credentials (e.g., board‑certified dermatologist, biomedical engineer) with affiliations and conflicts-of-interest statements.
- Use Organization and Person structured data to expose credentials (aligned with the on-page bios).
- Testimonials and before/after visuals:
- Disclose material connections, avoid misleading retouching, and indicate what’s typical when presenting results—per the FTC Endorsement Guides.
6) UX trust signals that move KPIs
- Security and identity: HTTPS, clear About and Contact, physical address, and customer support hours.
- Reviews with substance: Encourage detailed, experience-rich reviews (pros, cons, comparisons). This mirrors what Google encourages for product content in its ongoing Search Central Blog updates (Google, ongoing).
- Structured data: Product + Offer + AggregateRating/Review; Organization and Person. Align every claim in structured data with on-page text; no medical claims that your evidence/regulatory status doesn’t support.
- Public registry links: Offer verification links to FDA, NANDO, or safety alerts repositories (e.g., the EU’s Safety Gate portal, European Commission, ongoing) with neutral, non‑endorsement language.
- Monitor and respond: Watch MAUDE and recalls for your product and comparables; acknowledge lessons learned if relevant. Transparency after issues is a trust multiplier.
Context: Consumer trust correlates with purchase intent across categories. Annual research like the Edelman Trust Barometer (Edelman, 2019–2024) consistently links transparency and credible information with buyer confidence—even if precise DTC device uplift percentages aren’t publicly reported.
7) 2025: changes and common pitfalls to navigate
- MoCRA implementation: Safety substantiation, facility/product listings, serious adverse event reporting, and fragrance allergen rules are being operationalized. Use the FDA MoCRA hub (FDA, 2024–2025) to track updates and align your transparency page.
- EU Annex XVI CS: If you sell lasers/IPL or other Annex XVI products without medical purpose, make sure your EU pages reflect CS‑specific warnings, user training notes, and age restrictions—see EU 2022/2346 Common Specifications (EU, 2022).
- UK transition: Keep GB pages current with UKCA/CE acceptance timelines per MHRA guidance (updated 2024–2025).
Frequent errors I still see:
- “FDA approved” on 510(k) devices; solve with “510(k)-cleared” and a verification link to the FDA 510(k) database.
- CE logos on cosmetic creams; remove and reference EU cosmetics regulation and CPNP instead via the Commission’s CPNP page.
- Hand‑wavy specs: “clinical‑grade power” with no numbers. Replace with wavelength/frequency, dose, and tolerances.
- Unsubstantiated before/after photos or influencer claims that don’t reflect typical results—address via FTC health claims and endorsements guidance.
8) A 90‑day implementation plan that works
Days 0–15: Inventory and risk mapping
- List every device/SKU and market (US/EU/UK/other). Capture current claims, PDP specs, badges, and links.
- Determine intended use and regulatory status per market. Flag any claim that could trigger device/drug classification drift.
- Pull your standards/testing dossier: IEC/ISO, biocompatibility, EMC, clinical evidence.
Days 16–35: Content architecture and legal alignment
- Draft a sitewide Compliance hub structured by market, with placeholders for registry and guidance links (FDA, EU MDR/Annex XVI CS, MHRA).
- For each PDP, define a consistent “Regulatory Status,” “Key Specs,” and “Clinical Evidence” block.
- Write microcopy and disclaimers (e.g., “FDA registration does not imply approval”).
- Legal/Reg review for claims language; align with FTC health claims and endorsements guidance.
Days 36–60: Build, link, and ship
- Update PDPs with complete specs including dosage/parameters; upload IFU and spec PDFs with version control.
- Add verification links: FDA 510(k)/De Novo/PMA/Registration & Listing; NANDO; Safety Gate monitoring statement.
- Implement structured data: Product/Offer/Review; Organization/Person for brand and expert bios.
- Launch the Compliance hub with last‑updated stamps and a change log.
Days 61–90: Educate, test, and operationalize
- Publish a Knowledge Center article explaining your standards/testing approach, linking to IEC/ISO overviews like IEC 60825‑1 laser safety and IEC 60601‑1‑11 home‑use (IEC Webstore, previews) for context.
- A/B test PDP placement of “Regulatory Status” and “Key Specs” to measure engagement and conversion.
- Train customer support and influencers on compliant phrasing; update influencer briefs per FTC Endorsement Guides.
- Set a quarterly review to refresh links and address new guidance.
9) Copy blocks and micro‑templates you can reuse
Regulatory Status (US PDP example)
- “This device is 510(k)‑cleared for [indication]. Verify our clearance in the FDA’s 510(k) database under submission K######.”
- “Our facility is registered with the U.S. FDA. Registration does not imply product approval. Search our registration in the FDA Registration & Listing database.”
EU PDP warning (Annex XVI IPL example)
- “This product is an aesthetic device regulated under EU MDR Annex XVI and conforms to Common Specifications under Implementing Regulation (EU) 2022/2346. Read all warnings and training instructions before use. Not suitable for users under [age] or with [contraindications].”
Verification links block (Compliance page)
- “FDA 510(k) clearance: Search our model in the FDA 510(k) database.”
- “EU conformity assessment: Our Notified Body is listed in the NANDO database under [scope].”
- “Safety monitoring: We monitor FDA MAUDE and EU Safety Gate for safety alerts and recalls.”
10) Practitioner checklists
Product detail page (PDP) specs checklist
- [ ] Intended use/indications and target users
- [ ] Contraindications and key warnings
- [ ] Performance parameters (e.g., wavelength, fluence, frequency, current) with tolerances
- [ ] Photobiological/electrical safety standards referenced (e.g., IEC 62471, IEC 60601‑1/‑1‑2/‑1‑11)
- [ ] Biocompatibility approach (ISO 10993) and skin‑contact materials
- [ ] Electrical ratings, EMC classification, operating/environmental conditions
- [ ] Directions for use; maintenance/cleaning; troubleshooting
- [ ] Warranty terms and service contacts
- [ ] Downloadable IFU/spec sheet with version/date
- [ ] Regulatory Status block with appropriate verification links
Compliance hub checklist
- [ ] Market‑by‑market regulatory statements (US/EU/GB)
- [ ] Device classification/pathway and status
- [ ] Links: FDA 510(k)/De Novo/PMA; Registration & Listing; MAUDE; Recalls
- [ ] EU: Notified Body ID; NANDO link; Annex XVI CS notes
- [ ] GB: UKCA/CE acceptance statement with MHRA reference
- [ ] Cosmetics transparency (MoCRA): facility/product listing, safety substantiation, AE reporting, fragrance allergen plan with link to the FDA MoCRA hub
- [ ] Change log and “Last updated” timestamp
- [ ] Complaint handling and “Report an issue” channel
Claims substantiation file checklist (internal)
- [ ] Master list of all claims by SKU and region
- [ ] Evidence packets mapped to each claim (study design, endpoints, results, limitations)
- [ ] Testing reports (IEC/ISO, EMC, biocompatibility) with edition numbers
- [ ] Expert reviews and COIs; versioned bios
- [ ] FTC compliance notes for testimonials and visuals
- [ ] Audit trail for changes to marketing copy
11) How to talk about standards without overpromising
Keep it factual and scoped. Examples that consistently pass review:
- “Tested to IEC 62471 for photobiological safety; evaluated to IEC 60601‑1 and IEC 60601‑1‑2.”
- “Home‑use considerations addressed per IEC 60601‑1‑11.”
- “Skin‑contact materials assessed under ISO 10993 biocompatibility framework.”
For public education pages, you can reference standard overviews such as the IEC 60825‑1 laser safety listing (IEC Webstore) or CB explainer pages like Intertek’s home healthcare/IEC 60601‑1‑11 overview (Intertek) to help non‑engineers understand your testing scope.
12) Governance: keep E‑E‑A‑T alive after launch
- Quarterly reviews: Reconfirm registry links, refresh clinical summaries, and sync claim wording across PDPs, ads, and influencer briefs.
- Internationalization: Localize compliance pages per market; don’t reuse US claims on EU/GB pages if the regulatory status differs.
- Incident response: If a safety notice or recall occurs, add a dated statement on the Compliance hub with next steps and FAQs, and link to the relevant FDA/EU page (e.g., FDA recalls database).
- Schema hygiene: Update Organization/Person/Product structured data when experts change or when average ratings shift. Consistency is a trust signal.
Bottom line
In 2025, the most persuasive E‑E‑A‑T for beauty and personal‑care devices comes from disciplined transparency: precise claims, verifiable certifications, rigorous specs, and living compliance pages. Do this well and you’ll satisfy regulators, earn user trust, and give Google exactly what it’s looking for on YMYL pages—evidence of experience, expertise, and reliability.
Cited references for further action
- FDA’s central cosmetics modernization page: FDA MoCRA hub (2024–2025 updates)
- FDA device databases: 510(k) search • Registration & Listing • MAUDE
- EU regulations and databases: EU MDR 2017/745 consolidated • 2022/2346 Common Specifications • NANDO
- UK guidance: MHRA – Regulating medical devices in the UK
- Standards overviews: IEC 60601‑1‑11 preview • IEC 60825‑1 overview
- Advertising/claims: FTC health claims guidance (2023) • FTC Endorsement Guides (2023)
- Google quality resources: Helpful content guidance • Spam policies
- Trust context: Edelman Trust Barometer

